|
12/17/2023 0 Comments Periodic table chemistry class 
If Δχ is between 0.5 and 1.7, the bond is polar covalent. General Guidelines: If Δχ (difference in electronegativity) is less than 0.5, the bond is typically considered nonpolar covalent. Formula:Įlectronegativity Difference Method: It’s a simple method where the type of bond (ionic or covalent) is determined based on the difference in electronegativity values between the two bonded atoms. This scale is based on the average of the ionization energy (IE) and the electron affinity (EA) of an atom. Where E AA and E BB are the bond dissociation energies for the diatomic molecules A–A and B–B respectively. It’s based on the bond energies of different compounds.įormula: The difference in electronegativity between two atoms, A and B, can be related to the bond energy of the A–B bond, E AB, by the following equation: Linus Pauling introduced this widely used scale. Various scales have been developed to quantify electronegativity.Īlso Check – Molar Volume formula Pauling’s Scale It plays a fundamental role in understanding the type and nature of chemical bonds between atoms. 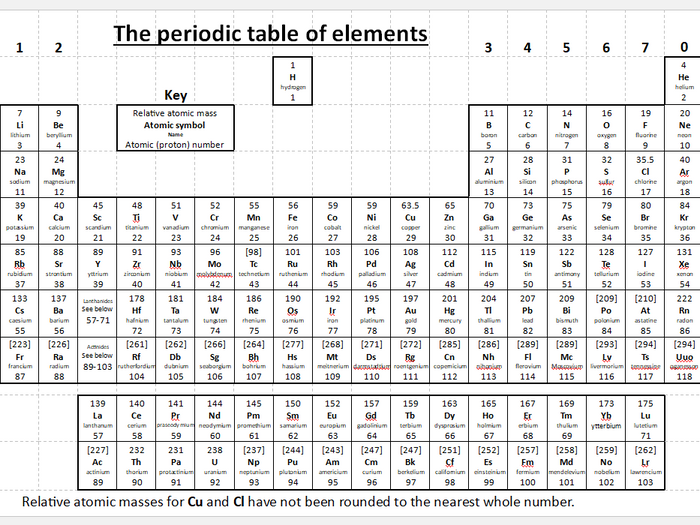
Note that while electron affinity is usually exothermic (releases energy), it can also be endothermic (requires energy) for some elements ElectronegativityĮlectronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. The energy for this process indicates the electron affinity. 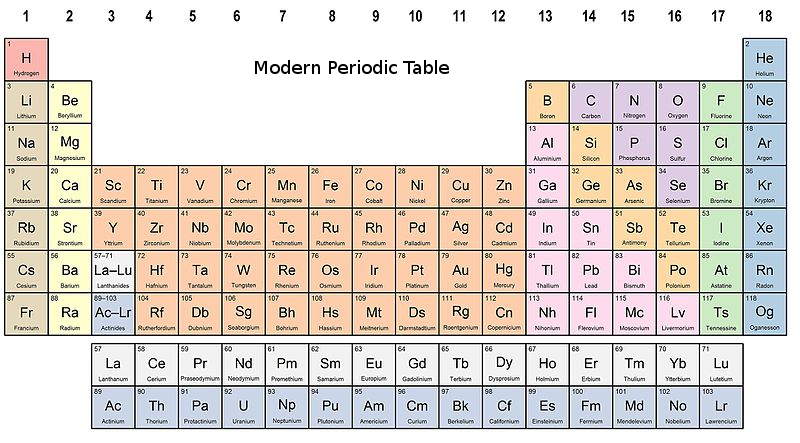
Equation: For a generic atom, B, the equation for electron affinity can be represented as: Electron Affinity (EA)ĭefinition: Electron affinity is the energy change when a neutral atom in the gaseous state accepts an electron to form a negatively charged ion. If we talk about the second ionization energy, it would be for the removal of another electron from A +, and so on for subsequent ionization energies. The energy for this process is the first ionization energy. Equation: For a generic atom, A, the equation for the first ionization energy is given by: Ionization Energy (IE)ĭefinition: Ionization energy is the energy required to remove the most loosely bound electron from a neutral atom in its gaseous state. P-block: Elements: Groups 13 to 18 (Boron, Carbon, Nitrogen, Oxygen, Halogens, and Noble gases) General electronic configuration: ns 2 np 1-6 “n” can be the second or greater principal quantum number.ĭ-block: Elements: Transition metals (Groups 3 to 12) General electronic configuration: (n-1) d 1-10 ns 0-2 The “d” orbitals being filled belong to an inner shell compared to the “s” orbitals.į-block: Elements: Lanthanides and Actinides General electronic configuration: (n-2) f 1-14 (n-1) d 0-1 ns 2 The “f” orbitals being filled are two shells inside compared to the “s” orbitals. S-block: Elements: Groups 1 and 2 (Alkali metals and Alkaline earth metals) General electronic configuration: ns 1-2 Where “n” is the outermost shell. Periodic classification is based on blocks and also provides trends in various atomic properties. 
“Periodic Table Formula: classification organizes chemical elements based on increasing atomic number, revealing recurring patterns and properties, grouped into periods and families.” What is the difference between ionization energy and electron affinity? Why do elements in the same group have similar chemical properties? How does metallic character vary across the periodic table? Why does atomic size generally decrease across a period? Hannay-Smith Electronegativity Difference Method.Pauling Electronegativity Difference Method.Each group is located in a different part of the periodic table. Elements are either metals, nonmetals, or metalloids (or semi metals). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
